Groundbreaking innovation combines SunTech‘s cuff-based blood pressure with Valencell’s PPG-based blood pressure monitoring technology
New technology set to transform patient safety, care, and comfort
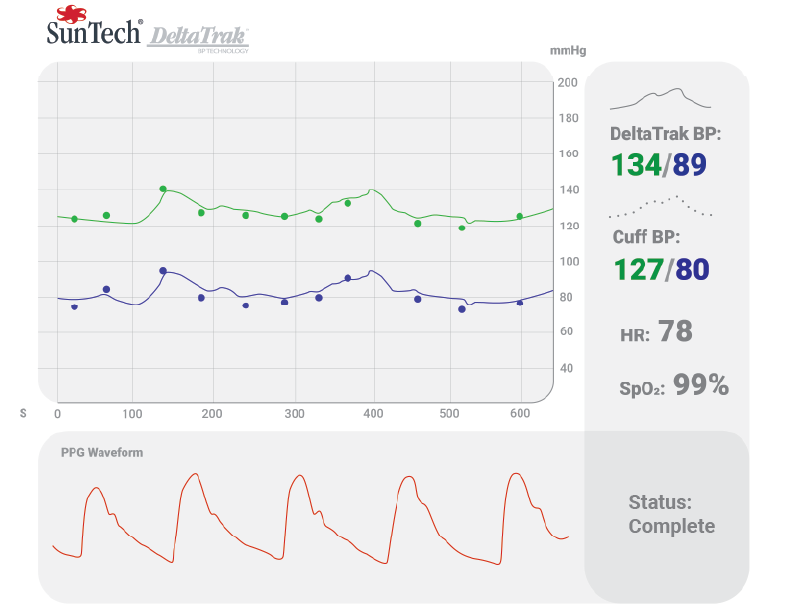
Morrisville, NC (February 8th, 2021) SunTech Medical Inc., the leading provider of clinical-grade blood pressure (BP) measurement technologies and devices, and Valencell Inc., the leader in highly accurate biometric sensors for wearables, today announce proof of concept for DeltaTrak™ technology. The patent-pending innovation combines the gold-standard accuracy of SunTech’s Advantage™ cuff-based BP and its superior motion tolerance capabilities, with continuous interpolative BP monitoring from Valencell’s optical PPG (photoplethysmography) technology between cuff readings. The proof of concept will allow for joint development discussions with medical device manufacturers interested in integrating this capability into their products.

Julian Mullaney, SunTech’s Vice President of R&D, commented: “The SunTech and Valencell collaboration has resulted in a groundbreaking solution that has not been possible before. We’re excited about the possibilities for this combination of motion-tolerant BP and PPG technology, particularly in the high-acuity clinical and veterinary market segments that can benefit from truly continuous monitoring.”
“Cuff BP measurements must be spaced apart in time to allow for blood flow, and patient comfort. The DeltaTrak™ technology is designed to provide comfortable BP monitoring continuously during and in between those clinical-grade cuff measurements,” explained Paul Matsumura, SunTech’s Sr. Director of Research and Technology.
DeltaTrak™ technology is designed to trigger a new cuff reading if the continuous signal violates pre-set guardrails. This functionality is intended to address the currently unmet need for continuous BP monitoring in situations where continuous cuff inflation is simply not feasible, such as hemodialysis, emergency medicine, OR and ICU, ambulatory BP studies, cardiac rehabilitation, and sleep. In clinical settings like sleep and hemodialysis, fewer cuff inflations could also provide a more comfortable patient experience.
 Dr. Steven LeBoeuf, President and Co-Founder of Valencell, commented: “The potential impact of this technology could be significant. SunTech and Valencell have developed a solution to an industry-limiting problem that has remained unsolved for decades – continuous BP monitoring combined with clinical-grade accuracy in high acuity situations. We hope this technology will address a critical unmet need.”
Dr. Steven LeBoeuf, President and Co-Founder of Valencell, commented: “The potential impact of this technology could be significant. SunTech and Valencell have developed a solution to an industry-limiting problem that has remained unsolved for decades – continuous BP monitoring combined with clinical-grade accuracy in high acuity situations. We hope this technology will address a critical unmet need.”
This innovation is the result of a multi-year collaboration between SunTech and Valencell, announced in January 2020. SunTech Medical’s parent corporation, Halma plc., is also an investor in Valencell and played an integral role in the collaboration.
“The cooperation with Valencell has been really productive,” said Rob Sweitzer, President of SunTech Medical. “Their data science team and knowledge of PPG tech, combined with SunTech’s experience in clinical markets and our BP algorithm experts, will result in valuable new solutions to improve patient safety.”
If you are interested in discussing the technology or to view a conceptual animated video of DeltaTrak™ BP, visit SunTech Medical at SunTechMed.com/DeltaTrak.
*DeltaTrak™ is not a product or medical device currently available for sale and has not been cleared by the FDA.
About SunTech Medical
SunTech Medical, a Halma company, is globally focused on the continual advancement of BP monitoring products and technologies for the benefit of our customers, patients, employees, and shareholders. Our passion is creating advances in clinical-grade BP technology and providing innovative products that improve the quality of patient care. Since the early 1980s, our sole focus, every day, has been on developing and improving leading-edge technology to obtain the most accurate and reliable BP measurements.
About Valencell
Valencell transforms the science of wearable biometrics to enable impactful health outcomes. Through innovative R&D and validated technologies, we develop breakthroughs and collaborate with wearables, hearables, and medical device companies around the world to deliver amazing results. Protected by more than 100 granted patents and more than 100 patents pending, Valencell’s technology can be used in wearables and hearables for virtually anyone, anywhere, doing anything. Valencell’s biometric sensor systems are currently integrated into more wearables, hearables and other form factors than any other technology provider in the world.
About Halma
Halma plc is a global group of life-saving technology companies. Our innovative products and solutions address many of the key issues facing the world today. Together, we have a purpose that drives everything we do: Growing a safer, cleaner, healthier future for everyone, every day.
Our purpose defines the three broad market areas where we operate:
- Safety: protecting life as populations grow and protecting worker safety.
- Environment: improving food and water quality, and monitoring air pollution.
- Health: meeting rising healthcare demand as growing populations age and lifestyles change.
They employ over 7,000 people in 20 countries, with major operations in the UK, Europe, the USA and Asia Pacific. Halma is listed on the London Stock Exchange (LON: HLMA) and is a member of the FTSE 100 index.
Media Contact:
Marissa Bellard
This email address is being protected from spambots. You need JavaScript enabled to view it.

